Quantification of polyreactive immunoglobulin G facilitates the diagnosis of autoimmune hepatitis.
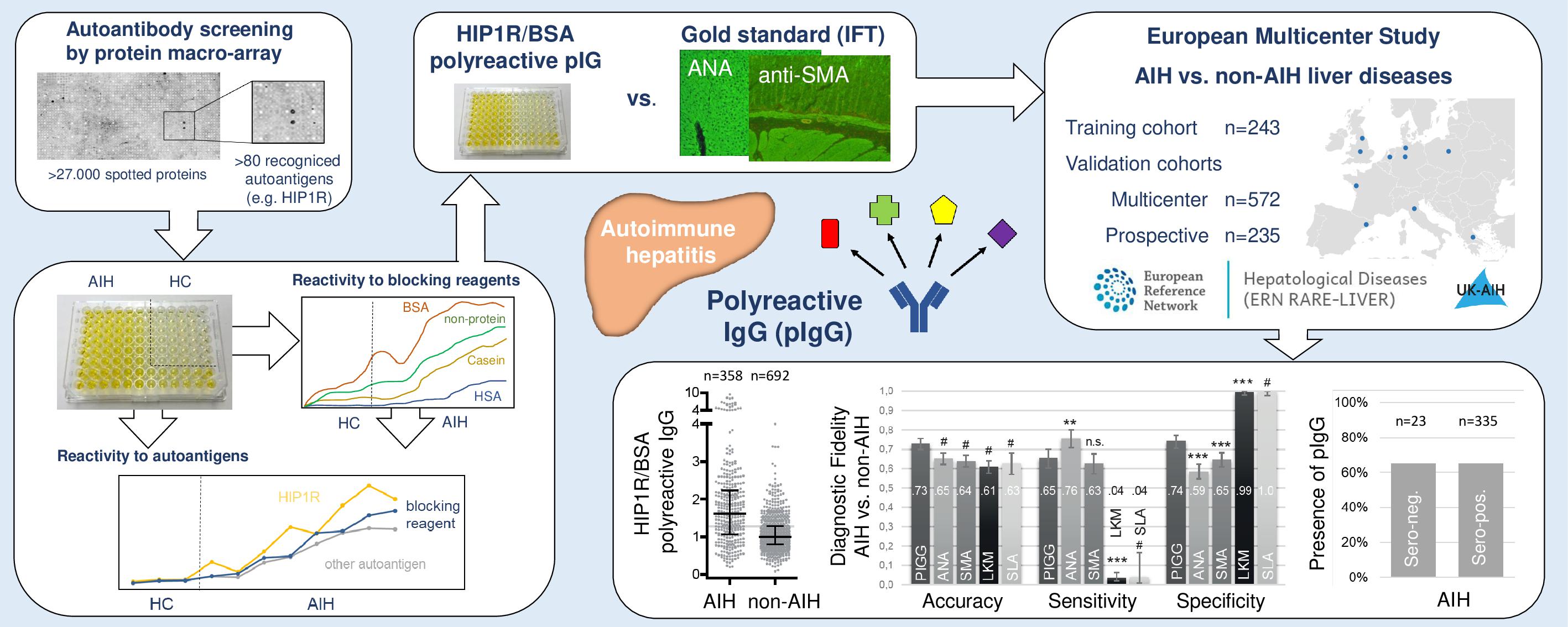
The authors, in collaboration with several colleagues from ERN RARE-LIVER and the International Au-toimmune Hepatitis Group (IAIHG), aimed to identify more accurate biomarkers for the diagnosis of AIH. Using a protein macro-array for initial identification and subsequent solid phase ELISAs for valida-tion, IgG antibodies that bound to many human and foreign proteins, including blocking reagents commonly used in immunoblots, were identified in AIH patients. Those polyreactive IgG (pIgG) were finally quantified by reactivity against human huntingtin-interacting protein 1-related protein in bovine serum albumin blocked ELISA (HIP1R/BSA). Three cohorts were recruited to assess the diagnostic ca-pacity of pIgG for the diagnosis of AIH. Within a retrospective training cohort from Hannover Medical School (MHH), a multicentric retrospective validation cohort from eight countries and a prospective validation cohort from MHH 1568 samples from adult patients were analyzed. pIgG had a 25% and 14% higher specificity to diagnose AIH than conventional anti-nuclear and anti-smooth muscle antibod-ies. pIgG was significantly more sensitive than anti-liver-kidney microsomal and anti-soluble liver anti-gen antibodies. Lastly, pIgG were 12-20% more accurate to diagnose AIH than conventional autoanti-bodies. In addition, pIgG were present in up to 71 % of AIH patients with normal IgG levels and in up to 88 % with seronegative AIH. If treated by immunosuppression, pIgG returned to background levels of non-AIH liver diseases. Therefore, pIgG might serve as a new biomarker for the diagnosis of AIH with a higher specificity and overall accuracy than conventional autoantibodies, and/or may be a good disease activity marker.